
While it was unclear at the time what the alpha particle was, it was known to be very tiny. Rutherford fired tiny alpha particles at solid objects such as gold foil. He found that while most of the alpha particles passed right through the gold foil, a small number of alpha particles passed through at an angle (as if they had bumped up against something) and some bounced straight back like a tennis ball hitting a wall. Rutherford performed a series of experiments with radioactive alpha particles. Rutherford proposes a planetary model of an atom In 1908, Ernest Rutherford, a former student of Thomson's, proved Thomson's raisin bread structure incorrect. These particles were later named electrons.Īfter Eugen Goldstein's 1886 discovery that atoms had positive charges, Thomson imagined that atoms looked like pieces of raisin bread, a structure in which clumps of small, negatively charged electrons (the "raisins") were scattered inside a smear of positive charges. Thomson theorized, and was later proven correct, that the stream was in fact made up of small particles, pieces of atoms that carried a negative charge. Thomson found that the mysterious glowing stream would bend toward a positively charged electric plate. For years scientists had known that if an electric current was passed through a vacuum tube, a stream of glowing material could be seen however, no one could explain why. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: the cathode ray tube.

Was not an "indivisible" particle as John Dalton had suggested but a jigsaw puzzle made of smaller pieces.
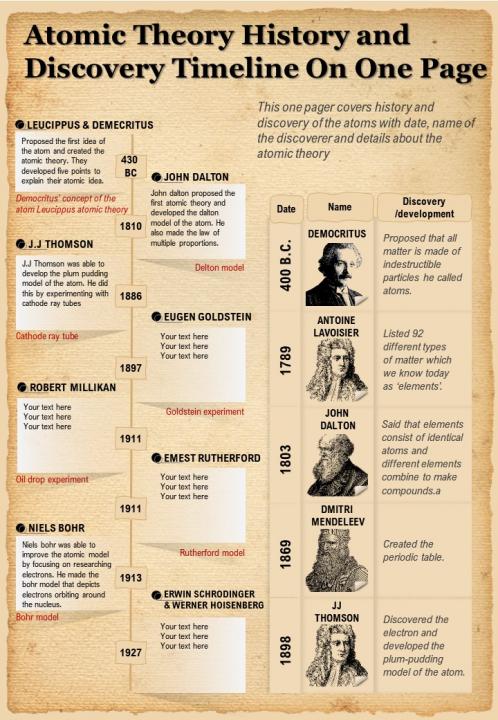
Thomson dramatically changed the modern view of the atom with hisĭiscovery of the electron. Until the final years of the nineteenth century, the accepted model of the atom resembled that of a billiard ball – a small, solid sphere. An updated version of the Atomic Theory I module is available.

